Starting in the era of Michael Faraday in the 19th century, people have long known that if there is a voltage on a metal surface and you place a solution of molecules above it, you can alter the molecules in a process called electrochemistry.
Unsurprisingly, this old practice has become artfully well developed over the years. Any chemist can tell you that if you take, for example, carbon dioxide, you can turn it into carbon monoxide using electrochemistry. However, it is still unclear what processes are actually going on right at the surface of the electrode when this happens.
There are experimental methods that can detect the miniscule, thin layers of surface molecules amidst the overwhelming quantities of non-surface molecules. These are readily applied to materials such as water or water-glass interfaces, yet it remains that very few experiments have been done that apply these methods to electrochemistry – despite there being a need for it in this area of research.
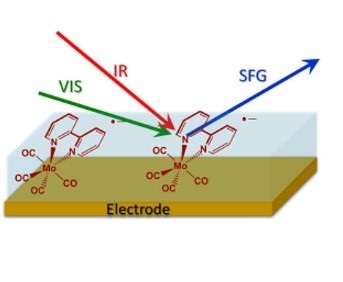
However, last year a team of 3 scientists from the University of Liverpool and the Central Laser Facility (Dr Alex Cowan (Liverpool), Dr Gaia Neri (Liverpool) and Dr Paul Donaldson (CLF)) used the Ultra laser facility to take a laser technique which is commonly used for surface observation and applied it to electrochemistry – a seemingly simple idea, but, if successful, would be a huge benefit to the electrochemistry community.
Using Ultra to make molecular level observations with the laser technique called IR-Visible vibrational sum frequency generation (VSFG), the technical outcome of this experiment was to observe the comings and goings of electrocatalyst molecules at metal electrodes and to find out why gold was better than platinum for an example of the CO2 to CO chemical reaction.