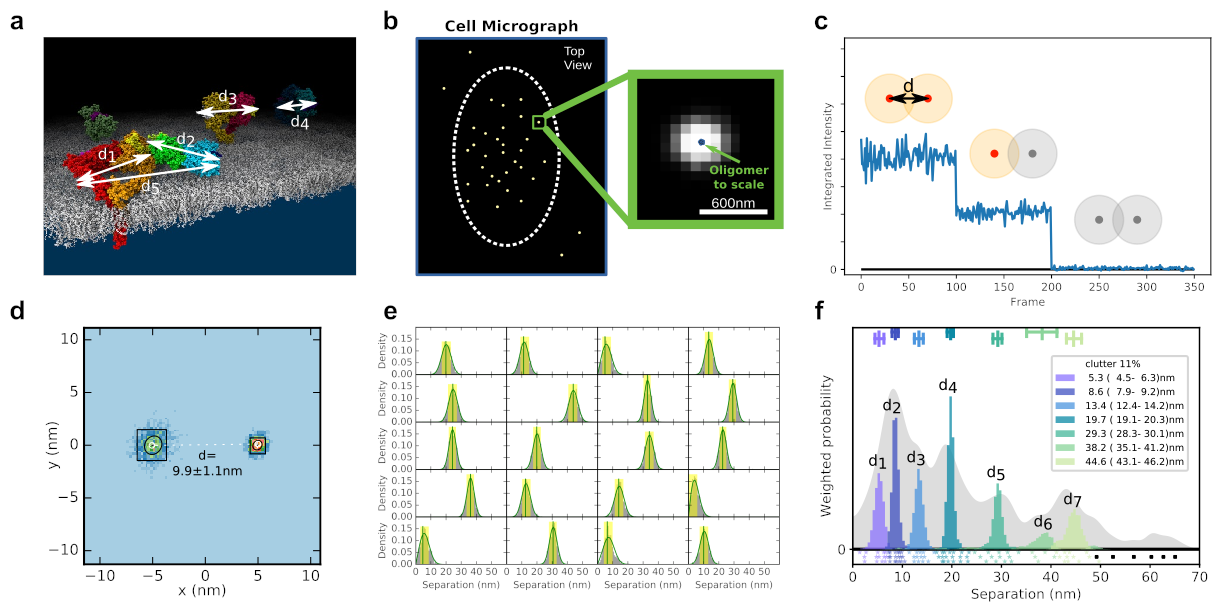
Overview of the technique
Fluorophore Localisation Imaging with Photobleaching (FLImP) is an end-to-end fully automated 2D single molecule imaging technique, which measures nanometre scale distances between fixed (stationary) fluorescently tagged proteins on the surface of cells (Figure A). It can resolve separations as small as 3nm using a single fluorophore.
In FLImP, samples are imaged using total internal reflection fluorescence (TIRF) microscopy that lets us see fluorescent tags attached to proteins close to the slide surface (typically <200nm, (Figure B). A short video is then recorded, over which time the fluorescent tags photobleach stochastically and single molecule tracking techniques are used to identify diffraction-limited spots where multi step photobleaching events have occurred. By counting the steps in the photobleaching staircase, we can estimate the number of fluorophores in each diffraction-limited spot (Figure C).
This information is used in conjunction with knowledge of point spread function shape to calculate the positions of multiple fluorophores within a single diffraction limited spot and so compute separations between them (Figure D). Separation sets from many such spots (Figure E) are then subject to Bayesian decomposition and the resulting distribution (a FLImP fingerprint, Figure F) reveals the set of discrete distances that describe how proteins are organised (Figure A).